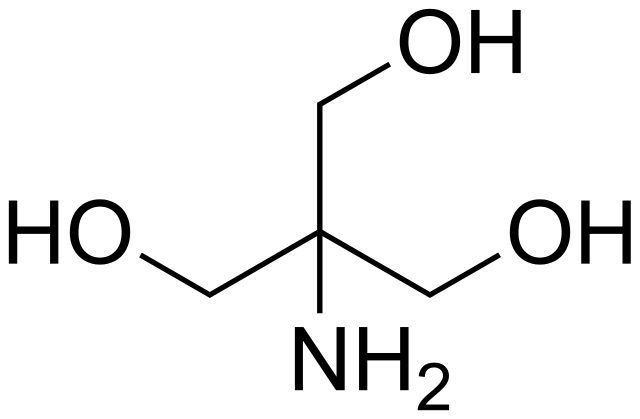
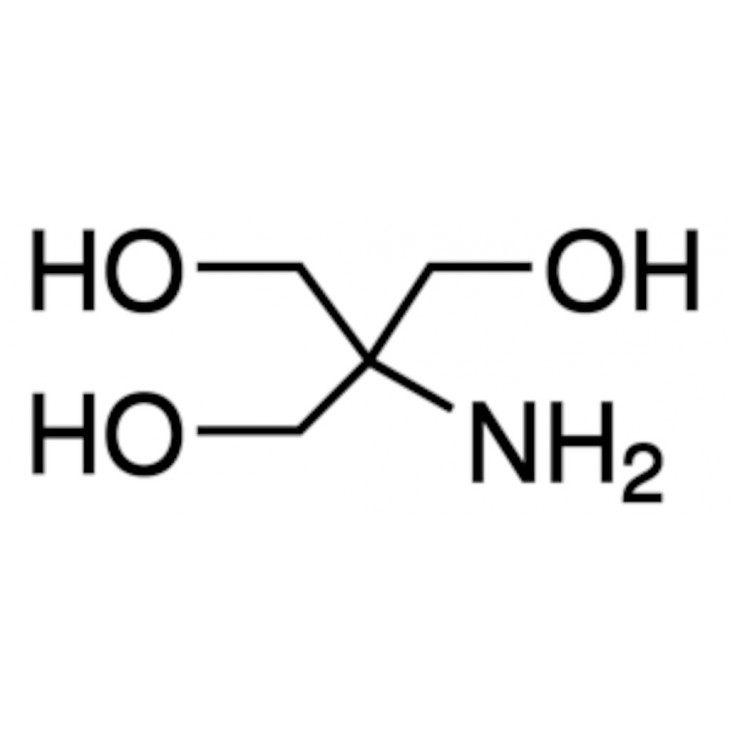
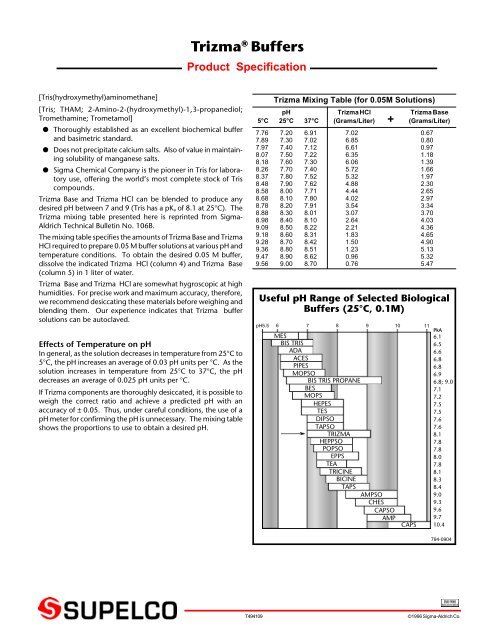
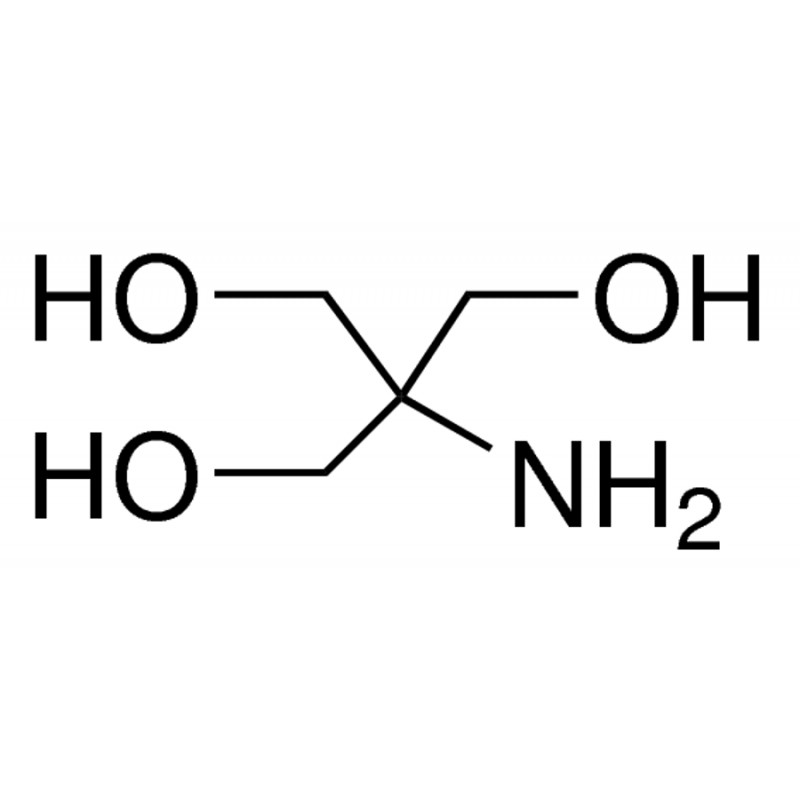
SOLVED: Tris' is a buffer often used in biochemistry. We can simplify the equilibrium reaction as: Tris-HCl + H2O -> Cl- + H3O+ + Tris with pKa = 8.072 and the molar

SOLVED: Tris(hydroxymethyl)aminomethane, abbreviated as Tris or TRIZMA, is frequently used to make buffers in biochemistry laboratories. Tris has a molecular weight of 121.1 g/mol, and its solution in water is basic with